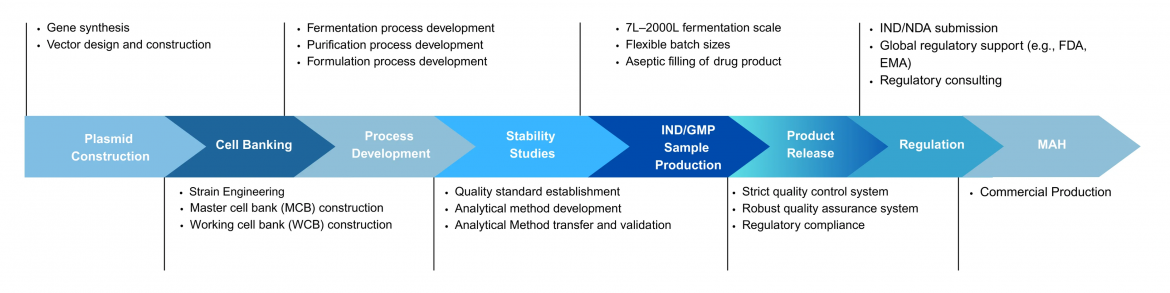
Ensuring the quality and safety of recombinant proteins is critical in the biotechnology sector. Good Manufacturing Practices (GMP) standards play a pivotal role in maintaining the integrity and efficacy of these products. As recombinant protein manufacturing becomes more prevalent in therapeutic applications, understanding GMP for recombinant proteins is essential for businesses in the life sciences field.
The Importance of GMP in Recombinant Protein Manufacturing
GMP guidelines are vital for ensuring that recombinant proteins are produced consistently and controlled to quality standards. These practices include maintaining sterile environments, conducting thorough quality control tests, and adhering to regulatory requirements set forth by agencies like the FDA and EMA. Compliance with GMP recombinant proteins standards not only safeguards the end user but also enhances a company’s reputation in the competitive market of therapeutic proteins.
The Role of Regulatory Bodies
Various regulatory bodies outline the framework for Yaohai Bio-Pharma GMP compliance in recombinant protein manufacturing. These organizations provide guidance on the design and operation of facilities, equipment qualifications, and validation of manufacturing processes. For companies developing GMP recombinant proteins, understanding these guidelines is crucial for navigating product approval pathways and ensuring successful market entry.
Future Trends in Recombinant Protein Production
The landscape of recombinant protein manufacturing is evolving with advancements in technology and methodologies. Companies are increasingly investing in automated processes and novel expression systems to enhance productivity and reduce costs. As demand for GMP recombinant proteins grows, it is essential for businesses to stay ahead of these trends, fostering innovation while ensuring compliance with stringent regulatory standards.
Partnering with Experts for Success
For businesses seeking to excel in recombinant protein manufacturing, partnering with experienced organizations such as Yaohai Bio-Pharma is invaluable. With a comprehensive platform for CMC research and GMP manufacturing services, Yaohai Bio-Pharma specializes in a broad range of recombinant proteins. Their expertise can guide businesses through the complexities of drug development, ensuring compliance with GMP standards while meeting diverse commercial needs. Embracing collaboration with leaders in the field can enhance product quality and accelerate time to market for recombinant protein therapies.